Abstract
Background: Essential thrombocythemia (ET) is a subtype of chronic myeloproliferative neoplasms (MPN) characterized by thrombocytosis and disease-related symptoms, which may be difficult to manage. Patients with ET are also at higher risk of thrombosis and hemorrhage. Ideal therapeutic approaches should achieve adequate cytoreduction, reduce the risk of thrombo-hemorrhagic complications, and prevent progression to post-ET myelofibrosis (PET-MF) or secondary acute myeloid leukemia (AML). Low-dose aspirin with hydroxyurea (HU) is typically given as first-line therapy in high-risk patients. However, approximately 20-40% of ET patients become HU-intolerant or -resistant. In ET, resistance and/or intolerance portend an increased risk of thrombosis, hemorrhage, disease transformation and death. There is a paucity of prospective clinical trial data to guide management of ET patients who are HU resistant or intolerant. P1101 is a next generation monopegylated interferon (IFN) alfa-2b, developed specifically to treat MPNs, including ET.
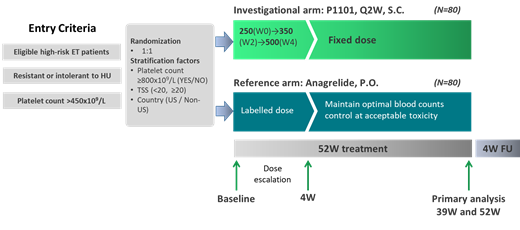
Study Design and Methods: The SURPASS-ET trial (NCT04285086) is a Phase 3, open-label, multicenter, randomized, active-controlled study to assess the efficacy, safety, tolerability, and pharmacokinetics (PK) of P1101 after 12 months of treatment compared with anagrelide as second line therapy for subjects with ET who have shown resistance or intolerance to HU. The primary endpoint is durable modified ELN composite response at 9 and 12 months from dosing. Cochran-Mantel-Haenszel test will be used for comparing the primary endpoint in the two treatment arms. The PK parameters of P1101, including (but not limited to) C min, T max, C max, and area-under-curve (AUC) will be derived using PPK analysis and the relationship between exposure and efficacy and safety endpoints will be examined using E-R analysis. Evaluation of efficacy will include clinical laboratory assessments, allelic burden measurements of CALR, JAK2, and MPL, spleen size measurements, bone marrow sampling (optional), EQ-5D-3L, and MPN Symptom Assessment Form Total Symptom Score (MPN-SAF TSS) assessments.
A total of 130 randomized subjects is planned to detect a difference of 40% (P1101) versus 15% (anagrelide) in durable ELN response rate with 90% power at alpha level = 0.05 using the chi-square test. To account for possible non-evaluability (e.g., no follow-up data), approximately 160 subjects will be randomized in this study to get 130 completed patients. Because of uncertainty in the assumptions on which the calculation of the sample size is based, an interim analysis for sample size adjustment will be implemented.
Major inclusion criteria include subjects diagnosed with high-risk ET (either older than 60 years and JAK2V617F-positive at screening or having disease-related thrombosis or hemorrhage in the past), diagnosed according to the World Health Organization (WHO) 2016 criteria, documented resistance/intolerance to HU and IFN naïve or anti-P1101 binding antibody negative. Key exclusion criteria include pregnant females, significant cardiovascular disease, documented autoimmune disease and a history or presence of clinically significant depression or neurological disease.
The study involves approximately 65 sites across the US, Taiwan, Japan, China, Hong Kong, Singapore, S. Korea, Canada, and Europe. To-date 55 patients (54 Asians, 1 Caucasian) have been randomized. The mean and median age at recruitment was 58.9 years (SD: 14.34) and 63 years (range 21 to 80 years) respectively. Twenty-seven men (49.1%) and twenty-eight women (50.9%) were recruited. Forty-two subjects (76.4%) had a TSS < 20. The study is being overseen by a Data Safety Monitoring Board (DSMB).
Mesa: Gilead: Research Funding; Promedior: Research Funding; AOP: Consultancy; Incyte Corporation: Consultancy, Research Funding; Abbvie: Research Funding; Genentech: Research Funding; La Jolla Pharma: Consultancy; Novartis: Consultancy; Sierra Oncology: Consultancy, Research Funding; Samus: Research Funding; Pharma: Consultancy; CTI: Research Funding; Celgene: Research Funding; Constellation Pharmaceuticals: Consultancy, Research Funding; CTI: Research Funding. Komatsu: Fujifilm Wako Pure Chemical Corporation: Research Funding; Fuso Pharmaceutical Industries, Ltd.: Research Funding; Japan Tobacco Inc.: Consultancy; Otsuka Pharmaceutical Co. Ltd: Membership on an entity's Board of Directors or advisory committees, Research Funding; Novartis Pharma KK: Consultancy, Research Funding, Speakers Bureau; Shire Japan KK: Consultancy, Research Funding, Speakers Bureau; PharmaEssentia Japan KK: Consultancy, Current Employment, Research Funding; Chugai Pharmaceutical Co., Ltd.: Research Funding; Kyowa Hakko Kirin Co., Ltd.: Research Funding. Sato: PharmaEssentia Japan KK: Current Employment. Qin: PharmaEssentia Corp.: Current Employment. Urbanski: PharmaEssentia Corporation: Current Employment. Shih: PharmaEssentia Corporation: Consultancy. Zagrijtschuk: PharmaEssentia U.S.A. Corp.: Current Employment. Zimmerman: PharmaEssentia Corporation: Current Employment. Verstovsek: Gilead: Research Funding; Protagonist Therapeutics: Research Funding; NS Pharma: Research Funding; Incyte Corporation: Consultancy, Research Funding; PharmaEssentia: Research Funding; Ital Pharma: Research Funding; CTI BioPharma: Research Funding; Blueprint Medicines Corp: Research Funding; AstraZeneca: Research Funding; Promedior: Research Funding; Genentech: Research Funding; Celgene: Consultancy, Research Funding; Roche: Research Funding; Novartis: Consultancy, Research Funding; Sierra Oncology: Consultancy, Research Funding; Constellation: Consultancy; Pragmatist: Consultancy.
Author notes
 This icon denotes a clinically relevant abstract
This icon denotes a clinically relevant abstract

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal